Electron Excitation and Emission
Electrons can absorb energy from external sources, such as lasers, arc-discharge lamps, and tungsten-halogen bulbs, and be promoted to higher energy levels. This tutorial explores how photon energy is absorbed by an electron to elevate it into a higher energy level and how the energy can subsequently be released, in the form of a lower energy photon, when the electron falls back to the original ground state.
In order to operate the tutorial, first choose an exciting wavelength by using the mouse cursor to translate the Wavelength (or Energy) slider to the desired position. Next, use the mouse to press the blue Pulse button, which will excite the atom by absorption of a photon of the chosen wavelength. Higher frequency wavelengths will elevate electrons in the atom to higher energy levels. After elevation to a higher energy state and circling the atom once, the electron will emit a photon of lower energy and decay to the ground state.
Visible light displays classical wave-like properties, but it also exhibits properties reminiscent of particles, which are manifested through entities that possess energy and momentum (but no mass), and are referred to as photons. The atom is the source of all forms of electromagnetic radiation, whether visible or invisible. Higher-energy forms of radiation, such as gamma waves and X-rays, are produced by events that occur to disrupt the nuclear stability of the atom. Radiation having lower energy, such as ultraviolet, visible, and infrared light, as well as radio and microwaves, originate from the electron clouds that surround the nucleus or the interaction of one atom with another. These forms of radiation occur due to fact that electrons moving in orbits around the nucleus of an atom are arranged in different energy levels within their probability distribution functions. Many of the electrons can absorb additional energy from external sources of electromagnetic radiation (see Figure 3), which results in their promotion to an inherently unstable higher energy level.
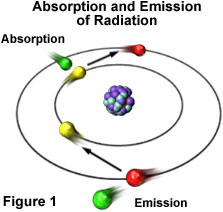
Eventually, the "excited" electron loses the extra energy by emitting electromagnetic radiation of lower energy and, in doing so, falls back into its original and stable energy level. The energy of the emitted radiation equals the energy that was originally absorbed by the electron minus other small quantities of energy lost through a number of secondary processes.
Electromagnetic radiation energy levels can vary to a significant degree depending upon the energy of source electrons or nuclei. For example, radio waves possess significantly less energy than do microwaves, infrared rays, or visible light, and all of these waves contain far less energy than ultraviolet light, X-rays, and gamma waves. As a rule, higher electromagnetic radiation energies are associated with shorter wavelengths than similar forms of radiation having lower energy. The relationship between the energy of an electromagnetic wave and its frequency is expressed by the equation:
where E is the energy in kilojoules per mole, h is Planck's constant, and the other variables are defined as discussed previously. Based on this equation, the energy of an electromagnetic wave is directly proportional to its frequency and inversely proportional to the wavelength. Thus, as frequency increases (with a corresponding decrease in wavelength), the electromagnetic wave energy increases, and vice versa.
